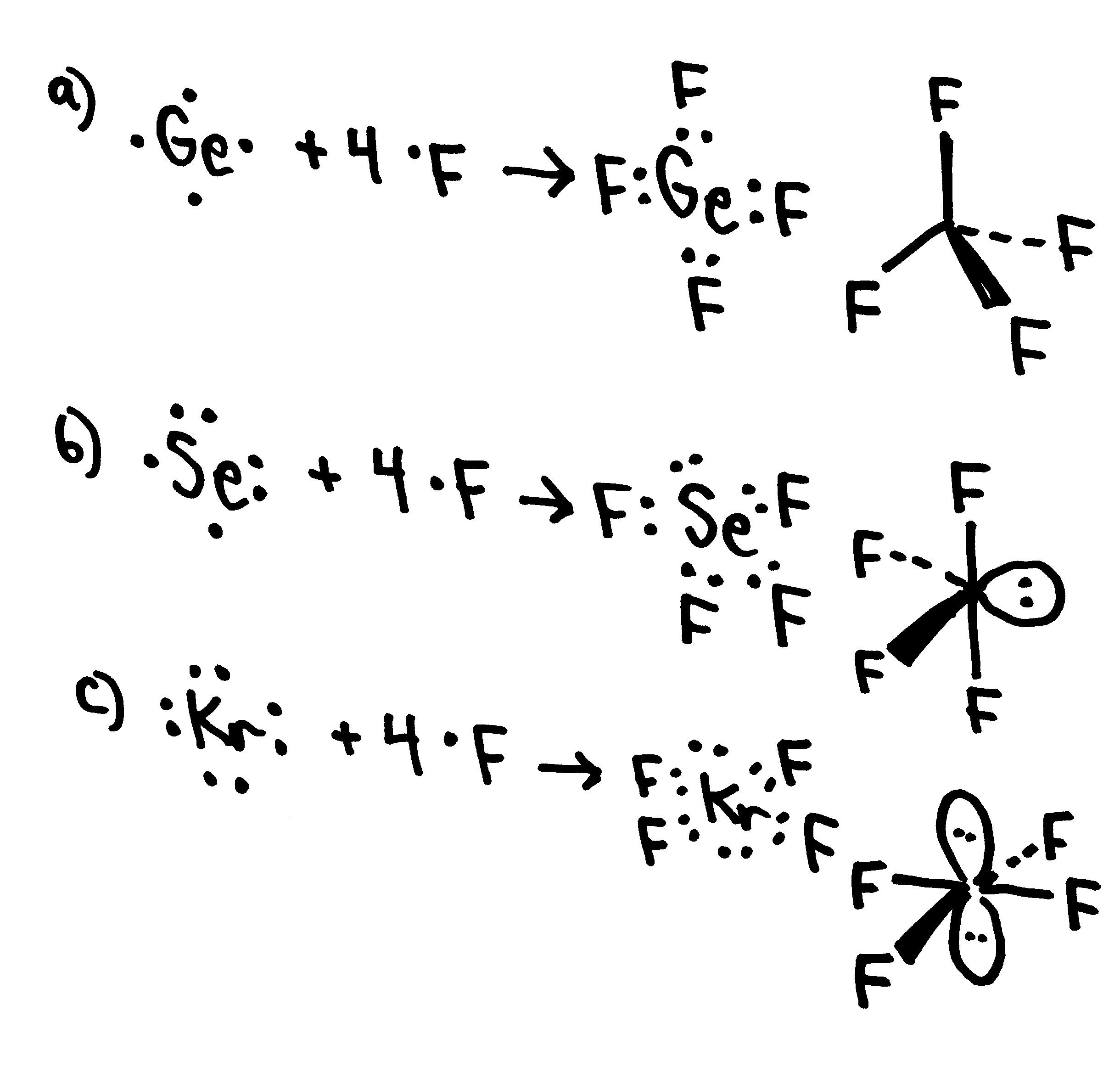
General Chemistry, chem 1b, 2nd midterm exam, spring, 20000
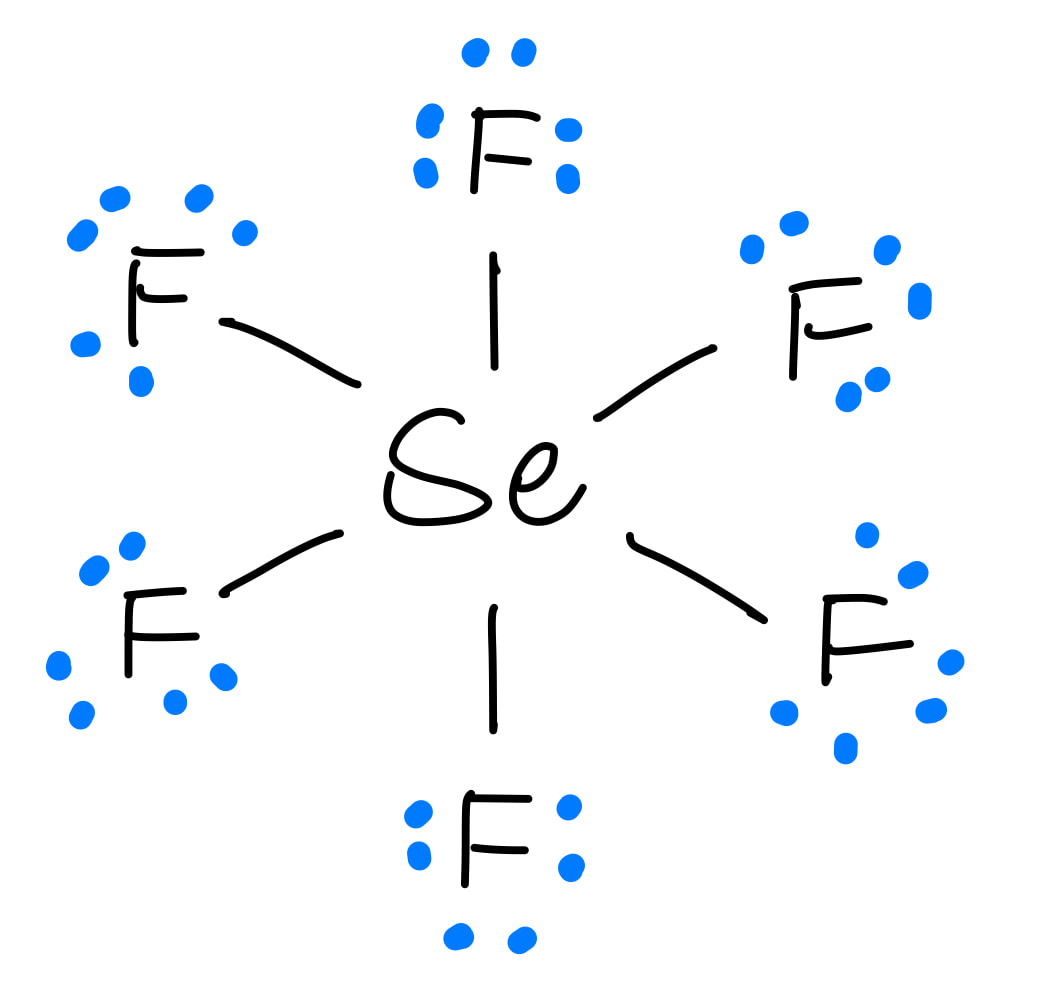
November 16, 2023 by Deep. The information on this page is fact-checked. Lewis structure of SeF 4. The Lewis structure of SeF4 contains four single bonds, with selenium in the center, and four fluorines on either side. There are three lone pairs on each fluorine atom, and one lone pair on the selenium atom. SeF4 Lewis Structure - How to Draw.

Xef4 Polar or Nonpolar KaileeropGuzman
Step 1: Count the total number of valence shell electrons on the compound Before drawing the structure, we need to know the number of valence shell electrons on all constituent atoms and their sum. Step 2: Draw the lewis dot structure for elements.
Lewis Structure For Sulfur
- Techiescientist Is SeF4 Polar or Non-Polar? SeF4, selenium tetrafluoride, is a colorless toxic and poisonous liquid that boils at a temperature close to that of water. Se has a +4 oxidation state in this molecule. Its molecular weight is 154.96 g/mol.
Ácido Fluorantimônico Química InfoEscola
SeF4 lewis structure is made up of one selenium and four fluorine atoms, selenium is the central atom, and fluorine is kept outside in the lewis diagram. There is one lone pair present on the central atom in the SeF4 lewis structure and 12 lone pairs on outer atoms. Follow some steps for drawing the lewis dot structure of SeF4 1.
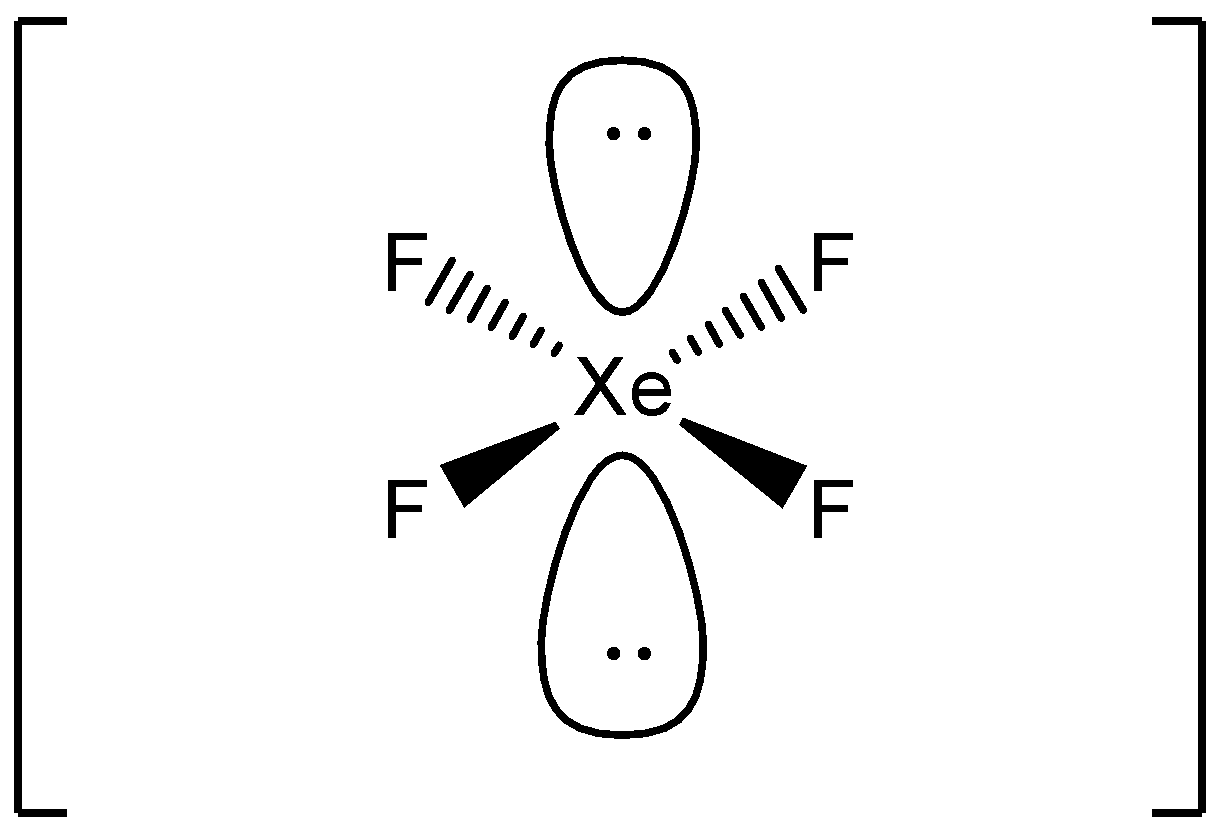
State true or false{{Xe}}{{{F}}_{{4}}} molecule is square planar in
Lewis structure of SeF4 contains four single bonds between the Selenium (Se) atom and each Fluorine (F) atom. The Selenium atom (Se) is at the center and it is surrounded by 4 Fluorine atoms (F). The Selenium atom has 1 lone pair while all the four fluorine atoms have 3 lone pairs. Let's draw and understand this lewis dot structure step by step.

SeF4 Lewis Structure How to Draw the Lewis Structure for SeF4 YouTube
An explanation of the molecular geometry for SeF4 (Selenium Tetrafluoride) including a description of the SeF4 bond angles. The electron geometry for the Sel.

Free download Lewis structure Sulfur hexafluoride Lewis pair VSEPR
Structural Formula. SeF 4. selenium tetrafluoride
VSEPR
In the SeF 4 Lewis structure, there are four single bonds around the selenium atom, with four fluorine atoms attached to it. Each fluorine atom has three lone pairs, and the selenium atom has one lone pair. Contents Steps #1 Draw a rough sketch of the structure #2 Next, indicate lone pairs on the atoms

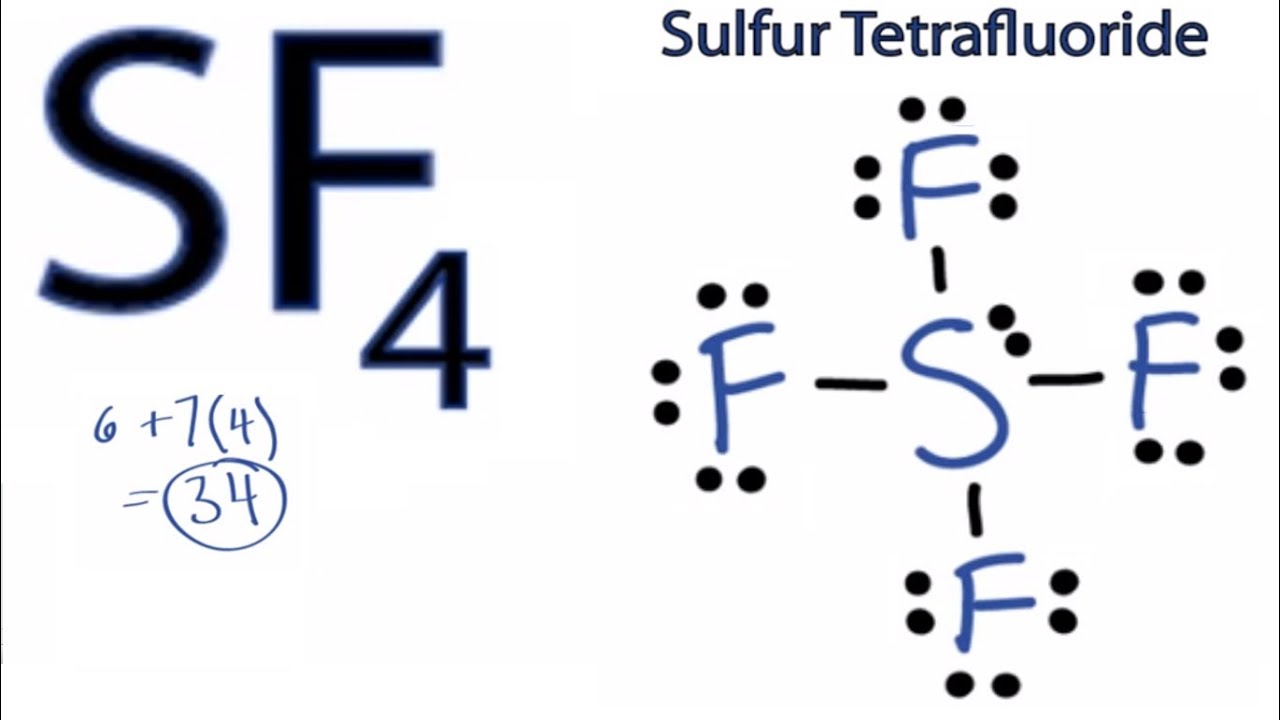
SF4 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram
Solution. Solutions to Example 10.4.1. Steps for Writing Lewis Structures. Example 10.5.1 10.5. 1. 1. Determine the total number of valence electrons in the molecule or ion. Each H atom (group 1) has 1 valence electron, and the O atom (group 16) has 6 valence electrons, for a total of 8 valence electrons.

SeF4 Molecular Geometry, Bond Angles (and Electron Geometry) YouTube
2 876 views 1 year ago Lewis Structure Hey there, are you looking for a video that can help you with SeF4 Lewis Structure? If yes, then you are at the right place! In today's video, we will.
Rncl2 Lewis Structure
Follow these simple steps to draw Lewis dot structures: Draw the atoms on paper and put dots around them to represent valence electrons of the atom. Be sure to have the correct number of electrons. If the species is an ion, add or subtract electrons corresponding to the charge of the ion.
SeF4 Lewis Structure, Geometry, Hybridization, and Polarity
SeF4 lewis structure has a Selenium atom (Se) at the center which is surrounded by four Fluorine atoms (F). There are 4 single bonds between the Selenium atom (Se) and each Fluorine atom (F). There is 1 lone pair on the Selenium atom (Se) and 3 lone pairs on all the four Fluorine atoms (F).

Chemfig How Can I Draw A Lewis Structure Tex Latex Stack Exchange Hot
Step 1/2. 1. SeF4: First, we need to determine the total number of valence electrons in SeF4. Selenium (Se) has 6 valence electrons, and each fluorine (F) atom has 7 valence electrons. Therefore, the total number of valence electrons in SeF4 is: 6 + 4 (7) = 34 Next, we arrange the atoms in a way that the central atom (Se) is surrounded by the.

Lewis Structure Sof4
A step-by-step explanation of how to draw the SeF4 Lewis Dot Structure (Selenium Tetrafluoride).For the SeF4 structure use the periodic table to find the tot.

Is Selenium tetafluoride (SeF4 )Polar or NonPolar? YouTube
The Lewis structure for SeF4 is written as: It can be seen in the structure that the octet for all the five atoms bonded to form the SeF4 molecule is satisfied i.e. the electronic configuration of all the four fluorine atoms as well as the selenium atom is eight. Steps of Drawing Lewis Structure of SeF4

SeF4 Lewis Structure (Selenium Tetrafluoride) YouTube
Solution. Steps for Writing Lewis Structures. Example 3.4.1 3.4. 1. 1. Determine the total number of valence electrons in the molecule or ion. Each H atom (group 1) has 1 valence electron, and the O atom (group 16) has 6 valence electrons, for a total of 8 valence electrons. 2.